Bromine is an active ingredient in four products; two products with multiple active ingredients and two products as the sole active ingredient. The multiple active ingredient products control mold, mildew, fungi, insects, and odors in exposed surfaces of bedding, mattresses, textiles, drapes, upholstered furniture, rugs, carpets, and storage areas. The electronegativity of Bromine is: χ = 2.96. In general, an atom’s electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity number, the more an element or compound attracts electrons towards it.
Answer to Write the electron configuration and give the orbital diagram of a bromine (Br) atom (Z = 35).
the σ bonds. I've drawn the overlaps below in the MO diagrams. Each bromine would donate one 4pz electron to form a σ -bonding orbital.
Answer to Draw an orbital diagram for each element:(a) magnesium; (b) aluminum; (c) bromine. wiringall.com!
Bromine atomic orbital and chemical bonding information. There are also tutorials on the first thirty-six elements of the periodic table.
Since bromine has 7 valence electrons, the 4s orbital will be (a)This diagram represents the correct filling of electrons for the nitrogen atom.The orbital diagram for Bromine is Share to: Orbital notation for beryllium?
chemistry is for lops Share to: What is the orbital notation of radon? [Rn]7s2 Share to: What is radon's orbital notation?
Electron configuration of radon is: [Xe]4f 14 5d 10 6s 2 6p 6. Answer to Write the electron configuration and give the orbital diagram of a bromine (Br) atom (Z = 35).
Note that in linear diatomic molecules, the p_z orbital always points along the internuclear axis, so it has to contribute to one of the sigma bonds. I've drawn the overlaps below in the MO diagrams.
BROMINE BONDING (HOMONUCLEAR DIATOMIC) For 'Br'_2, it is the simpler of the two examples. Electron Configurations and Orbital Diagrams KEY Draw orbital diagrams for the following elements: 1.
phosphorus ↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑ ↑ ↑ 1s 2s 2p 3s 3p 4s 3d 4p. Mar 22, · Best Answer: Yes; bromine (atomic number 35) has 1 less electron than the next higher inert gas, krypton (atomic number 36) so its electron configuration is: [Ar],3d10,4s2,4p5 Hope this answers your wiringall.com: Resolved.Orbital-Filling Diagram for Bromine | Chemistry Learning Apps | Pat ThayerOrbital filling diagram for Bromine?
| Yahoo Answers
How many electrons are there in #Br^-#?
Bromine Electrons Lost

1 Answer
Explanation:
The atomic number for bromine is 35, which means it has 35 protons in its atomic nuclei. A neutral bromine atom would also have 35 electrons. In order for a bromine atom to become a
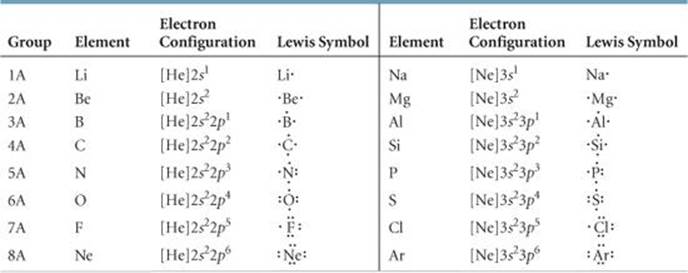
Below is the Lewis dot structure for a neutral bromine atom, which has seven valence electrons.
Below is the Lewis dot structure for a
Bromine Electron Shell
The diagram below shows how a bromine atom gains an electron from the element lithium in order to form the ionic compound LiBr.
Bromine Electrons Neutrons Protons
Related questions
