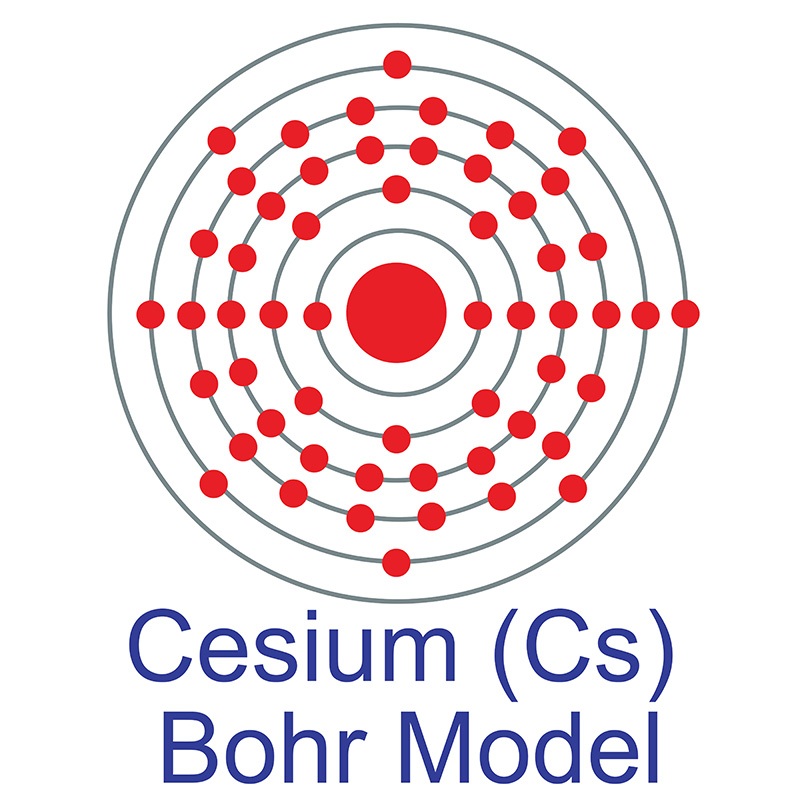
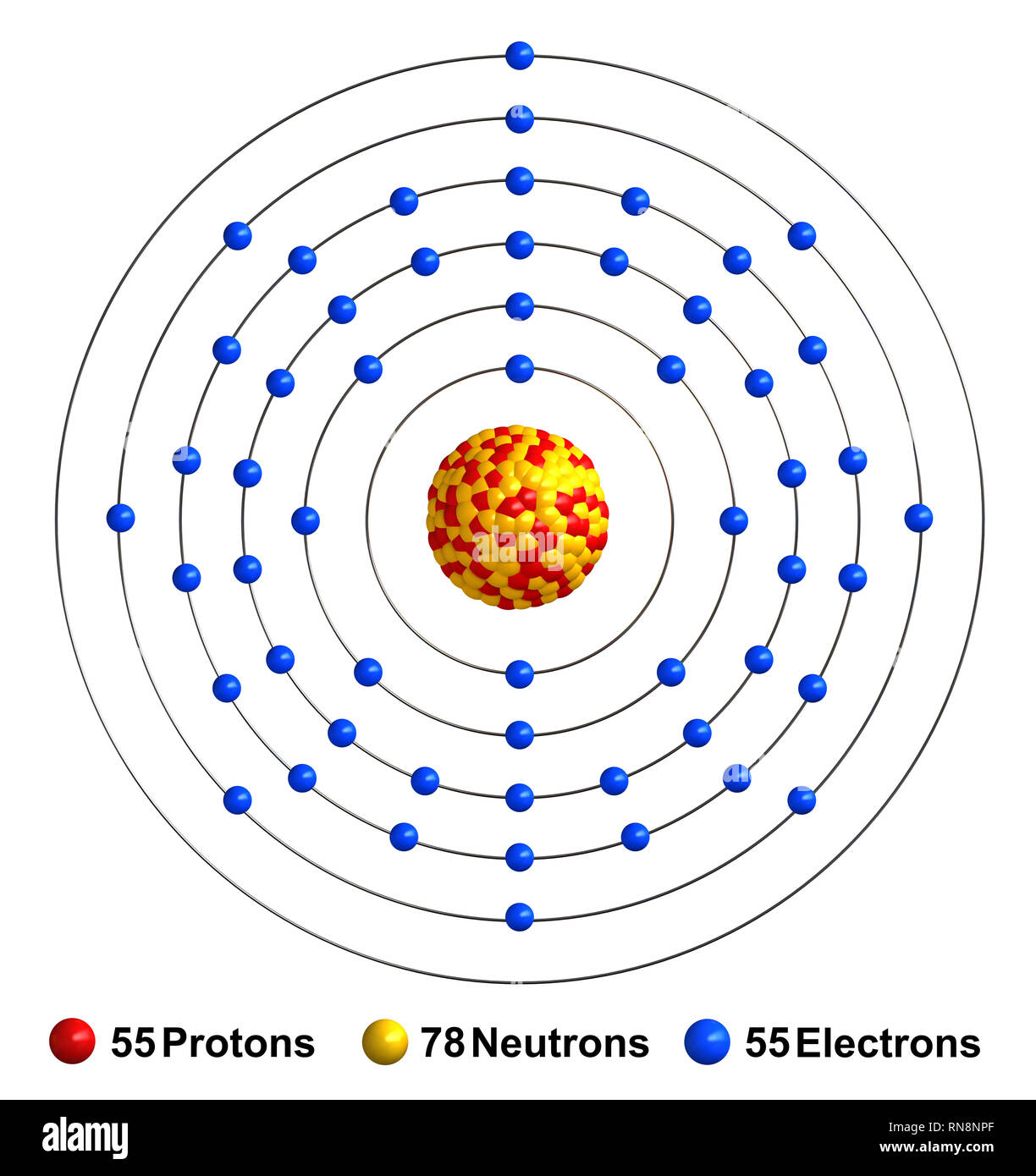
To find out the atomic number of cesium, we can use the periodic table. With the help of the periodic table, we can easily see that the atomic number of cesium is 55. As its atomic number is 55, it has a total of 55 protons, and for neutral cesium, the number of protons is always equal to the number of electrons i.e. 55 electrons in the nucleus. Caesium (IUPAC spelling) (also spelled cesium in American English) is a chemical element with the symbol Cs and atomic number 55. It is a soft, silvery-golden alkali metal with a melting point of 28.5 °C (83.3 °F), which makes it one of only five elemental metals that are liquid at or near room temperature. Element Caesium (Cs), Group 1, Atomic Number 55, s-block, Mass 132.905. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images.
- Cesium-137 Part 1 – Radioactive Material Identification Common Names: Cesium-137 Chemical Symbol: Cs-137 or 137Cs Atomic Number: 55 Mass Number: 137 (82 neutrons) Chemical Form: Cesium chloride Physical Form: A pellet of cesium ceramic housed in a welded.
- Name: Cesium Symbol: Cs Atomic Number: 55 Atomic Mass: 132.90546 amu Melting Point: 28.5 °C (301.65 K, 83.3 °F) Boiling Point: 678.4 °C (951.55005 K, 1253.12 °F) Number of Protons/Electrons: 55 Number of Neutrons: 78 Classification: Alkali Metal Crystal Structure: Cubic Density @ 293 K: 1.873 g/cm 3 Color: silver British Spelling: Caesium.
Overview
Cesium is a member of the alkali family, which consists of elements in Group 1 (IA) of the periodic table. The periodic table is a chart that shows how chemical elements are related to each other. The alkalis include lithium, sodium, potassium, rubidium, and francium. Cesium is considered the most active metal. Although in theory francium is more active than cesium, francium is too rare to have any commercial uses.
Cesium was discovered in 1861 by German chemists Robert Bunsen (1811-99) and Gustav Kirchhoff (1824-87). They found the element using a method of analysis they had just invented: spectroscopy. Spectroscopy is the process of analyzing light produced when an element is heated. The light produced is different for every element. The spectrum (plural: spectra) of an element consists of a series of colored lines.
SYMBOL
Cs
ATOMIC NUMBER
55
ATOMIC MASS
132.9054
FAMILY
Group 1 (IA)
Alkali metal
PRONUNCIATION
SEE-zee-um
Cesium is not a common element, and it has few commercial uses. One of its radioactive isotopes, cesium-137, is widely used in a variety of medical and industrial applications.
Discovery and naming
The invention of spectroscopy gave chemists a powerful new tool. In many cases, the amount of an element present in a sample is too small to see. But the element is much easier to detect by spectroscopy. When the substance is heated, the hidden elements give off characteristic spectral lines. Using spectroscopy, a chemist can identify the elements by these distinctive lines.
Such was the case with the discovery of cesium. In 1859, Bunsen and Kirchhoff were studying a sample of mineral water taken from a spring. They saw spectral lines for sodium, potassium, lithium, calcium, and strontium. These elements were already well known.
After Bunsen and Kirchhoff removed all these elements from their sample, they were surprised to find two beautiful blue lines in the spectrum of the 'empty' spring water. The water contained an unknown element. Bunsen suggested calling the element cesium, from the Latin word caesius for 'sky blue.' For many years, the name was also spelled caesium.
Physical properties
Cesium is a silvery-white, shiny metal that is very soft and ductile. Ductile means capable of being drawn into thin wires. Its melting point is 28.5°C (83.3°F). It melts easily in the heat of one's hand, but should never be handled that way! Cesium's boiling point is 705°C (1,300°F), and its density is 1.90 grams per cubic centimeter.
Chemical properties
Cesium is a very reactive metal. It combines quickly with oxygen in the air and reacts violently with water. In the reaction with water, hydrogen gas is released. Hydrogen gas ignites immediately as a result of the heat given off by the reaction. Cesium must be stored under kerosene or a mineral oil to protect it from reacting with oxygen and water vapor in the air.
Cesium also reacts vigorously with acids, the halogens, sulfur, and phosphorus.
Occurrence in nature
The abundance of cesium in the Earth's crust has been estimated at about 1 to 3 parts per million. It ranks in the middle
of the chemical elements in terms of their abundance in the earth.Cesium occurs in small quantities in a number of minerals. It is often found in an ore of lithium called lepidolite. The mineral containing the largest fraction of cesium is pollucite (Cs 4 Al 4 Si 9 O 26 ). This ore is mined in large quantities at Bernic Lake, in the Canadian province of Manitoba. Cesium is also found in small amounts in a mineral of boron called rhodizite.
Isotopes
Cesium Atomic Number And Weight
Only one naturally occurring isotope of cesium is known, cesium-133. Isotopes are two or more forms of an element. Isotopes differ from each other according to their mass number. The number written to the right of the element's name is the mass number. The mass number represents the number of protons plus neutrons in the nucleus of an atom of the element. The number of protons determines the element, but the number of neutrons in the atom of any one element can vary. Each variation is an isotope.
A number of artificial radioactive isotopes of cesium are known also. A radioactive isotope is one that breaks apart and gives off some form of radiation. Radioactive isotopes are produced when very small particles are fired at atoms. These particles stick in the atoms and make them radioactive.
One radioactive isotope of cesium is of special importance, cesium-137. It is produced in nuclear fission reactions. Nuclear fission is the process in which large atoms break apart. Large amounts of energy and smaller atoms are produced during fission. The smaller atoms are called fission products. Cesium-137 is a very common fission product.
Nuclear fission is used in nuclear power plants. The heat produced by nuclear fission can be converted into electricity. While this process is going on, cesium-137 is being produced as a by-product. That cesium-137 can be collected and used for a number of applications.
As an example, cesium-137 can be used to monitor the flow of oil in a pipeline. In many cases, more than one oil company may use the same pipeline. How does a receiving station know whose oil is coming through the pipeline? One way to solve that problem is to add a little cesium-137 when a new batch of oil is being sent. The cesium-137 gives off radiation. That radiation can be detected easily by holding a detector at the end of the pipeline. When the detector shows the presence of radiation, a new batch of oil has arrived.
This isotope of cesium can also be used to treat some kinds of cancer. One procedure is to fill a hollow steel needle with cesium-137. The needle can then be implanted into a person's body. The cesium-137 gives off radiation inside the body. That radiation kills cancer cells and may help cure the disease.
When a hollow steel needle filled with cesium-137 is implanted into a person's body, the isotopes's radiation can kill cancer cells.
Cesium-137 is often used in scientific research also. For example, cesium tends to stick to particles of sand and gravel. This fact can be used to measure the speed of erosion in an area. Cesium-137 is injected into the ground at some point. Some time later, a detector is used to see how far the isotope has moved. The distance moved tells a scientist how fast soil is being carried away. In other words, it tells how fast erosion is taking place.
Cesium-137 has also been approved for the irradiation of certain foods. The radiation given off by the isotope kills bacteria and other organisms that cause disease. Foods irradiated by this method last longer before beginning to spoil. Wheat, flour, and potatoes are some of the foods that can be preserved by cesium-137 irradiation.
Extraction
Cesium can be obtained in pure form by two methods. In one, calcium metal is combined with fused (melted) cesium chloride:
In the other, an electric current passes through a molten (melted) cesium compound:
Uses
Cesium has a limited number of uses. One is as a getter in bulbs and evacuated tubes. The bulb must be as free from gases as possible to work properly. Small amounts of cesium react with any air left in the bulb. It converts the gas into a solid cesium compound. Cesium is called a getter because it gets gases out of the bulb.
Cesium is also used in photoelectric cells, devices for changing sunlight into electrical energy. When sunlight shines on cesium, it excites or energizes the electrons in cesium atoms. The excited electrons easily flow away, producing an electric current.
An important use of cesium today is in an atomic clock. An atomic clock is the most precise method now available for measuring time. Here is how an atomic clock works:
Cesium-137 is used in atomic clocks, the most precise method for measuring time.
A beam of energy is shined on a very pure sample of cesium-133. The atoms in the cesium are excited by the energy and give off radiation. That radiation vibrates back and forth, the way a violin string vibrates when plucked. Scientists measure the speed of that vibration. The second is officially defined as that speed of vibration multiplied by 9,192,635,770.
Atomic clocks keep very good time. The best of them lose no more than one second in a million years.
Compounds
Cesium compounds have relatively few commercial uses. Cesium bromide is used to make radiation detectors and other measuring devices. Cesium carbonate and cesium fluoride are used to make specialty glasses. Cesium carbonate and cesium chloride are used in the brewing of beers. Cesium compounds are also used in chemical research.
Cesium Barium Atomic Number
Health effects
Cesium is not regarded as essential to the health of plants or animals, nor does it present a hazard to them.
Under standard conditions, the single caesium or cesium body is a soft, ductile metal, white or silver to gold. Its melting point 28.5 °C (83.3 °F) is close to ambient and human body temperatur, at which it can remain in a liquid state by supercooling; cesium shares this property with gallium and rubidium, mercury being the only pure metal that is liquid and evaporates at room temperature.
This chemical element is a soft, golden-white alkali metal, which is one of the five metallic elements that is liquid at or around room temperature. The most famous use of this chemical element is in atomic clocks.
Cesium is the most alkaline of the alkali metals (strongest base known; enough to attack glass). Its chemical properties are close to those of rubidium and potassium, which belong to the same family. It reacts violently with water and air (explosive reaction). It is extremely reactive and pyrophoric, and reacts with water even at temperatures as low as −116° C.
It is extracted from pollucite (aluminosilicate Cs4Al4Si9O26·H2O) but it is also present in trace amounts in lepidolite.
The use of caesium
In medicine, as a source of radioactive radiation to treat cervical cancer and bladder cancer.
Cesium chloride is now used as a remedy in alternative medicine. This use follows the theory linking the progression of cancerous metastases to intracellular pH. Thus, this alkaline element enters the cancer cell through Na + K + ATPase and blocks the glucose trying to enter through the plasma membrane. The daily dose is 0.5 to 3 grams of cesium chloride in solution to create the saturation effect lethal to fermenting cancer cells.
Cesium chloride is now used as a remedy in alternative medicine. This use follows the theory linking the progression of cancerous metastases to intracellular pH. Thus, this alkaline element enters the cancer cell through Na + K + ATPase and blocks the glucose trying to enter through the plasma membrane. The daily dose is 0.5 to 3 grams of cesium chloride in solution to create the saturation effect lethal to fermenting cancer cells.
In atomic clocks, the use of cesium-133 achieves an accuracy of one second for 1.6 million years. This element is also used in centrifugation and in photomultipliers. Cesium formate is used in drilling to form a denser mud which promotes the rise of crushed rock. Cesium 137 is an indicator of the fallout from nuclear tests and accidents.
Photomultipliers (device that converts photons into electrons, then multiplies them so as to obtain a signal in the form of a current): these vacuum tubes use the emission by a photocathode, then the multiplication of electrons by a succession of electrodes (dynodes) – up to 12 or 14. The photocathode frequently includes cesium compounds, especially in the infrared. Compared to solid-state detectors, the photomultiplier is much more bulky and delicate to use.
Caesium 137 toxicity and effect on health
Produced artificially by nuclear activities, cesium 137 has an impact on health. It is radiotoxic to the liver and kidneys and also appears to increase the occurrence of cardiovascular disease. Cesium 137 may interfere with skeletal development upon in utero exposure in rats. Vitamin D metabolism is also affected under these conditions.
A distinction is generally made between external exposure and internal exposure to cesium 137. Skin contaminations are difficult to eliminate (the period in the skin being about four days) and a high local dose of cesium 137 causes a skin burn. which should be treated as a classic burn.
Why is Caesium 137 so dangerous? Cesium-137 is an especially dangerous fission product because of its high yield during fission, moderate half-life, high-energy decay pathway, and chemical reactivity. Because of these properties, cesium-137 is a major contributor to the total radiation released during nuclear accidents.
Internal exposure is much more dangerous. Indeed, if it is inhaled or ingested, cesium-137 is assimilated like its counterpart and natural competitor, potassium, throughout the body by concentrating preferentially in the muscles (with a greater load in the child than in adults). Its biological half-life (that is, the time after which half of the cesium-37 that has entered the body is released to the outside in the urine, stools and sweat) is approximately 100 days.
The ingestion of large doses of cesium 137 has devastating effects: bone marrow failure, impairment of the immune system and reproductive function, kidney diseases, etc. In addition, at lower doses and in the longer term, cesium 137 causes an increase. thyroid cancer, congenital and fetal malformations, as well as neurological disorders.
In terms of the therapeutic management of patients contaminated with cesium 137, Prussian blue (iron ferrocyanide) is the only effective treatment for evacuating cesium 137 after ingestion.
Caesium in the periodic table
| Atomic number (Z) | 55 |
|---|---|
| Group | group 1: H and alkali metals |
| Period | period 6 |
| Block | s-block |
| Electron configuration | [Xe] 6s1 |
| Electrons per shell | 2, 8, 18, 18, 8, 1 |
Physical properties
| Physical properties | |
|---|---|
| Phase at STP | solid |
| Melting point | 301.7 K (28.5 °C, 83.3 °F) |
| Boiling point | 944 K (671 °C, 1240 °F) |
| Density (near r.t.) | 1.93 g/cm3 |
| when liquid (at m.p.) | 1.843 g/cm3 |
| Critical point | 1938 K, 9.4 MPa[2] |
| Heat of fusion | 2.09 kJ/mol |
| Heat of vaporization | 63.9 kJ/mol |
| Molar heat capacity | 32.210 J/(mol·K) |
Vapour pressure
| P (Pa) | 1 | 10 | 100 | 1 k | 10 k | 100 k |
|---|---|---|---|---|---|---|
| at T (K) | 418 | 469 | 534 | 623 | 750 | 940 |
Other properties
| Other properties | |
|---|---|
| Natural occurrence | primordial |
| Crystal structure | body-centred cubic (bcc) |
| Thermal expansion | 97 µm/(m·K) (at 25 °C) |
| Thermal conductivity | 35.9 W/(m·K) |
| Electrical resistivity | 205 nΩ·m (at 20 °C) |
| Magnetic ordering | paramagnetic[4] |
| Young’s modulus | 1.7 GPa |
| Bulk modulus | 1.6 GPa |
| Mohs hardness | 0.2 |
| Brinell hardness | 0.14 MPa |
| CAS Number | 7440-46-2 |
History
| History | |
|---|---|
| Naming | from Latin caesius, sky blue, for its spectral colours |
| Discovery | Robert Bunsen and Gustav Kirchhoff (1860) |
| First isolation | Carl Setterberg (1882) |
Main isotopes of caesium
| Main isotopes of caesium | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||
Periodic Table of Elements | Complete List of Chemical Elements by Group, Name, Symbol, Color and Type

Sources: Royal Society of Chemistry, Web MD, Live Science
Photo credit: Wikimedia Commons
Photo explanations: Pollucite, a caesium mineral. An example of the mineral Pollucite on display in the Vale Inco Limited Gallery of Minerals at the Royal Ontario Museum.
